In Vitro Fertilization (IVF)
Although IGO no longer operates an IVF laboratory or program as such, we work closely with area SART‐ reporting clinics to offer our infertility patients continuity of care. IGO patients who need IVF will be referred to one of these clinics for their IVF cycle stimulation, egg retrieval and embryo transfer. All preliminary testing done at IGO will be provided to the IVF clinic. After completion of the IVF cycle, IGO welcomes the opportunity to provide follow‐up care to our patients whether this entails continued infertility or gynecologic care, and, of course, obstetric care when a successful pregnancy is achieved.
In Vitro Fertilization (IVF)
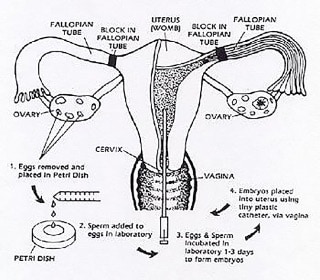
In Vitro Fertilization, originally developed to treat infertility caused by blocked or damaged fallopian tubes, is currently used to treat a variety of infertility problems. With IVF the eggs and sperm are collected and then mixed together in a laboratory dish so that fertilization occurs outside the body. Several days later the developing embryo is transferred to the woman's uterus where implantation can occur and result in a pregnancy.
IVF can be used to treat many infertility problems either present singly or in combination. These include:
- Tubal factor (blocked or damaged fallopian tubes)
- Ovulation problems
- Cervical factor
- Endometriosis
- Male factor (low sperm count, poor motility, poor morphology)
- Immunological factor (antisperm antibodies in either partner) Idiopathic infertility (infertility of unknown cause)
There are four basic steps to the IVF process. The first is ovarian stimulation to facilitate the production of multiple mature eggs. Stimulation involves the use of fertility drugs (e.g. Pergonal®, Repromex ®, Metrodin®, Fertinex®, Gonal‐F® and others) which act on the ovary to cause many follicles to mature instead of only the one which typically matures in an unstimulated cycle. Ultrasound measurements and blood hormone levels are checked frequently to carefully monitor the follicle development, to determine the subsequent fertility drug dose, and to time the retrieval of the mature eggs. The amount of fertility drugs needed for a stimulation cycle depends on the woman's age and her fertility diagnosis. When the oocytes (eggs) are mature, they are retrieved by ultrasound guided follicle aspiration. This procedure is performed under local anesthesia in the operating room. A vaginal ultrasound probe is used to guide a needle through the vaginal wall, into the ovary and to the follicles. One‐by‐one the follicles are aspirated and the resulting fluid is taken to the ART laboratory to identify and recover the oocytes.
In the ART laboratory, under very carefully controlled conditions, the oocytes are identified and described as to their maturity and appearance. They are then transferred to specially designed culture media and placed in an incubator. A semen specimen is produced by the male and processed by laboratory personnel to obtain the strongest, most active sperm in the ejaculate. Prepared sperm are mixed with the eggs and placed in the incubator under conditions controlled for temperature and humidity in a culture media designed to mimic the environmental conditions of the female's fallopian tube at the time of ovulation. After 16 to 20 hours, the eggs are inspected to detect signs of fertilization (pronuclear formation); after 48 hours, if embryo development is occurring normally, the embryo is at the two to six cell stage. At 72 hours (three days), the embryo is at eight or more cells. By five to six days, the embryo will be at the blastocyst stage if development is occurring as expected.

Embryos may be transferred to the uterus on day three or later at the blastocyst stage. The timing of transfer is determined by the number and development of the embryos, the patient's age, and by decision of the physician and the patient couple. Embryos are placed in a tiny, flexible catheter and transferred to the uterus by the physician. During a pelvic exam, the catheter is threaded through the cervix into the uterus and the embryos are very carefully expelled in a small volume of media. Embryo transfer is not a complicated procedure and is performed without anesthesia. If the IVF cycle is successful, the embryo(s) will begin to implant in the uterine wall within a few days and a pregnancy test will be positive about 10 to 12 days after the embryo transfer. Two to three weeks later an ultrasound study will be done to confirm the presence of an intrauterine pregnancy.
Blastocyst Culture
To improve the chances of a successful pregnancy and reduce the obstetrical risks associated with multiple births, much emphasis has been placed on improving laboratory culture techniques. Recent advances in culture media allow embryologists to grow embryos to the blastocyst stage(five to six days instead of two to three days before transfer). Because theembryo is allowed to grow longer, only the strong survive (those with a better chance of causing a full‐term pregnancy). Therefore, only one, twoor three blastocysts are transferred to the uterus.
This new technology does not mean that everyone gets a happy ending. Blastocyst culture often does not work well for older women because fewer of their eggs will produce embryos that are strong or healthy enough make it to blastocysts, and these women typically have fewer eggs at the start. These older women and other women who produce fewer eggs risk not having a blastocyst embryo transfer because no embryos survive to this stage of development. In addition, because fewer embryos survive for all women (due to the natural selection process for healthy strong embryos), there will be fewer excess embryos to be frozen for future use even with the youngest women.
Couples need to be aware that by using the blastocyst culture technique, we can select the healthiest embryos with the greatest chance of achieving a pregnancy for transfer; however we risk not having an embryo transfer (or having only one embryo to transfer) and we risk not having embryos to cryopreserve for future use. For the typical patient, if sufficient eggs and developing embryos are available (five or more), we will generally recommend blastocyst culture and transfer. As with all the infertility treatments at IGO Medical Group, the specifics of any couple's care is tailored to their individual needs and, very importantly, the couple is encouraged to be actively involved in the decision process.
Intracytoplasmic Sperm Injection (ICSI)
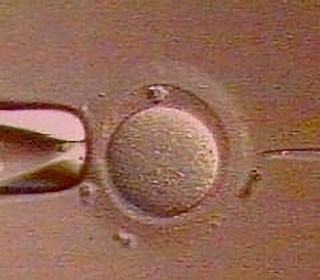
The ICSI procedure is performed during an in vitro fertilization cycle on the day of oocyte retrieval. Using mechanically controlled microscopic instruments under a microscope, a single sperm cell is directly injected into the cytoplasm (interior) of the oocyte (egg). The sperm must be processed prior to injection in order to select the morphologically best sperm for use. The oocyte must be processed prior to injection in order to remove the surrounding outer layers of cells and to assess the oocyte to be sure it is at the proper developmental stage for fertilization to occur.
The oocyte's outer protective layer (the zona pellicuda) and the cell membrane are pierced by a very fine glass pipette and the sperm cell is deposited directly inside the oocyte. This process does not guarantee that fertilization will occur, only that the sperm is placed in the appropriate location for fertilization. To achieve fertilization, the sperm head must undergo a process (decondensation) and the oocyte must be "activated" in order for male and female pronuclei to form (the proof that fertilization has occurred).
Intracytoplasmic sperm injection (ICSI) is used in clinical situations where a male factor infertility problem is known or strongly suspected. Clinical indications for the use of ICSI include very low motile sperm number, very poor motility characteristics, a significantly high number of morphologically abnormal sperm, the presence of a high concentration of antisperm antibodies, and/or the use of sperm derived surgically from the epididymis or testes. These indications for ICSI are not all inclusive or "set in stone". The recommendation to use ICSI in your clinical setting will be determined on an individual basis by you, your physician, the laboratory director and embryologists.
Intracytoplasmic Sperm Injection differs from conventional IVF in two specific ways. With conventional IVF, rather than injecting the sperm directly into the oocyte, many sperm are placed in the culture media with the oocyte and then the gametes are incubated overnight in the hopes that the sperm will enter the egg "naturally".
Assisted Hatching (AH)
The Assisted Hatching procedure is performed during an IVF treatment cycle just prior to the embryo transfer. Using mechanically controlled microscopic instruments and an enzymatic solution, a tiny opening is created in the zona pellucida (the outer layer covering over the oocyte) on the day of embryo transfer (at the cleaving embryo stage). In theory, the creation of this opening will facilitate the hatching of the developing blastocyst (embryo) from the zona, a process that is necessary before the embryo can implant in the uterine wall.
Assisted Hatching (AH) is used in several clinical settings and worldwide it is used extensively. For our purposes, indications for its use include female patient age greater than 36 years old, a female patient baseline FSH value greater than 10 in our assay, increased zona pellucida thickness as determined by microscopic observation, embryos created by ICSI (defined above) and transferred on day 3, patients who have previously undergone IVF and had morphologically good embryos but no resulting pregnancy, and patients with embryos that appear to develop at a slower rate than expected. These indications are not all inclusive or "set in stone". The recommendation to use AH in your clinical situation will be determined on an individual basis by you, your physician, the laboratory director and embryologists. When Assisted Hatching is used, it is done on the day of embryo transfer. The embryos are assisted hatched an hour or two prior to embryo transfer. If the embryos are cultured to blastocysts (day 5 to 7), Assisted Hatching is not typically done; however, certain clinical situations may suggest that AH would be beneficial.
Donor Eggs for IVF
Donor eggs can be used for IVF in situations where the woman's eggs are not suitable. This alternative is appropriate when the woman carries a genetic disorder that she does not want to pass on to a child, when she has premature ovarian failure and her ovaries are not longer capable of being stimulated to produce mature eggs, when she has undergone repeated IVF cycles without success or when increased age (in her 40's) increases the chances of egg abnormalities interfering with successful pregnancy. Egg donors can be anonymous or known to the couple. They are usually matched to the woman for ethnic background and physical characteristics as decided by the infertile couple. Donors are carefully screened to, as carefully and as thoroughly as possible, rule out genetic abnormalities, medical conditions, sexually transmitted diseases and psychological issues that would make them unsuitable as an egg donor.
IVF using an egg donor is essentially the same as conventional IVF except that the ovaries are stimulated and the eggs are retrieved from the donor and inseminated with the husband's sperm in the laboratory; the resulting embryos are transferred to the wife's uterus in the hopes of achieving a pregnancy. This means that the egg donor's and the wife's cycles must be synchronized such that the wife's uterus is receptive to the embryos shortly after the egg donor produces mature eggs. If synchronization is not successful in any given IVF attempt, all embryos can be cryopreserved and used by the couple in a later, more suitable, cycle.
Egg donor IVF can be a very realistic means for an older woman to achieve pregnancy. Fairly high pregnancy rates have been reported since the eggs used are typically from young, pregnancy‐proven donors.
Embryo Cryopreservation and Frozen Embryo Transfer
Embryos produced in excess of those used for transfer in IVF can be cryopreserved for future use. The embryos are mixed with a special solution, placed in small storage straws and frozen using a very specific, controlled rate freezing chamber. Cryopreserved embryos are then stored in liquid nitrogen tanks and remain viable for several years. Embryos can be thawed and used for embryo transfer in future cycles. Using this technique, a couple can get more than one pregnancy attempt from a single ovarian stimulation and egg retrieval event. The method and solutions used minimize the risk of cell damage during the freeze or thaw processes. From 50 to 75% of the cryopreserved embryos will survive the thaw process and be available for embryo transfer.
Pre‐implantation Genetic Diagnosis
Pre‐implantation genetic diagnosis (PGD) is a technique that can be used during IVF procedures to test embryos for genetic disorders prior to their transferring into the uterus. The procedure involves removing (biopsying) one or two cells from an embryo by a micro‐manipulation procedure. The biopsied cell(s) are fixed on a slide and several of the chromosomes stained to determine whether an abnormal number of these chromosomes is present (e.g. Trisomy 21, Down’s syndrome). For some genetic diseases in which the single gene causing the disease has been identified (e.g. cystic fibrosis), the DNA of the embryo can be examined to see if the genetic condition exists in that embryo.
